MTAA Forums and Working Groups
The role of Forums and Working Groups is to provide an opportunity for interaction between Members with a specific area of common interest.
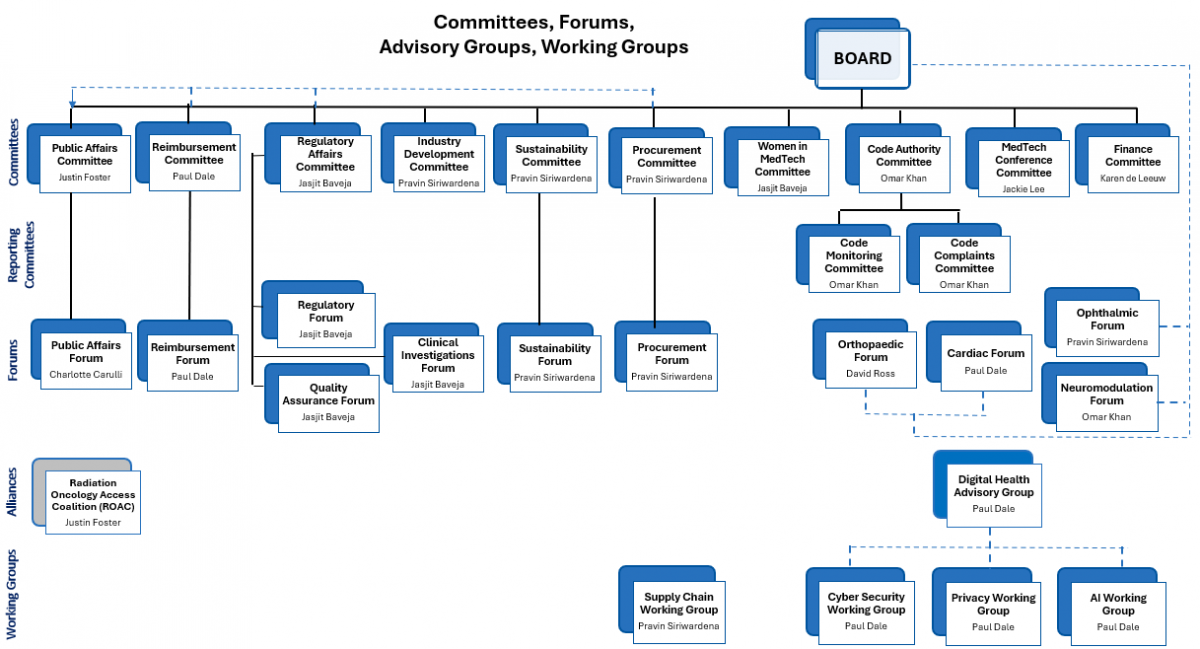
Forums
Cardiac Forum
Currently – Prostheses List reform in relation to cardiac implantable electronic devices, engagement with Cardiac Society. Future topics TBD.
Neuromodulation Forum
Representative forum for MTAA neuromodulation member companies. Develop policy and advocacy strategies pertaining to neuromodulation products, including on the Prosthesis List; engage with neuromodulation industry stakeholders
Ophthalmic Forum
Representative forum for MTAA ophthalmic member companies. Develop policy and advocacy strategies pertaining to intraocular lens products, including on the Prosthesis List; engage with ophthalmic industry stakeholders
Orthopaedic Forum
The MTAA Orthopaedic Forum is a representative forum of MTAA member companies with a special interest in matters relating to the manufacture and supply of orthopaedic devices, such as:
- Prostheses List reforms specifically applicable to orthopaedic implants
- Publication of Special Access Scheme Category C List for Joint Implant Components no Longer in the ARTG
- MTAA Orthopaedic Market Survey, covering hips, knees, shoulders, spinal, trauma, sports implants and bone cement which has 17 participating companies. Aggregated survey data is updated twice per year and is available only to participating companies
- Matters of special interest involving MTAA and the Australian Orthopaedic Association are discussed quarterly between the two associations
- The MTAA Orthopaedic Forum meets quarterly
Procurement Forum
Development of guiding principles for industry procurement; develop and sustain relationships with procurement officials / category managers at state level; determine solutions to offer state officials to harmonise procurement processes
Regulatory Forum
The Regulatory Affairs Forum is an extension of the Regulatory Affairs Committee (member base is large and covers a range of members), and acts as an information-sharing space to ensure the broad membership is covered and issues are passed up to the committee and subsequently to the TGA and RegTech Committee.
Sustainability Forum
The Forum has been established to inform, navigate and lead members regarding activities in the evolving policy and regulatory landscape in relation to environmental, social, and governance (ESG).
Clinical Investigations Forum (CIF)
The Clinical Investigations Forum comprises of MTAA member companies who conduct Clinical Trials in Australia. The forum allows important information to be shared to ensure arising issues are identified and actioned through the MTAA Industry Policy Committee and the Research & Development Task Force, and acts as an educational conduit amongst the broader MTAA membership.
Interest / Working Groups
The Digital Healthcare Advisory Group is comprised of MTAA members and non-members (including representatives from academic institutions, the Australasian Telehealth Society etc.) and aims to become a partner with the Australian Government to improve digital healthcare in Australia and to develop advocacy and policy positions relevant to MTAA members that manufacture medical devices with monitoring capabilities. Click here to read more.
Cyber Security Working Group
Exclusively focused on cyber security and privacy issues affecting the medtech industry.
Privacy Working Group
AI Working Group
Research & Development Task Force
This Task Force is a joint membership of MTAA, MA and AusBiotech members with the goal to work with Government on their various initiatives and to shape the Clinical Trials space in Australia to ensure Australia becomes a preferred location to conduct Clinical Trials.
